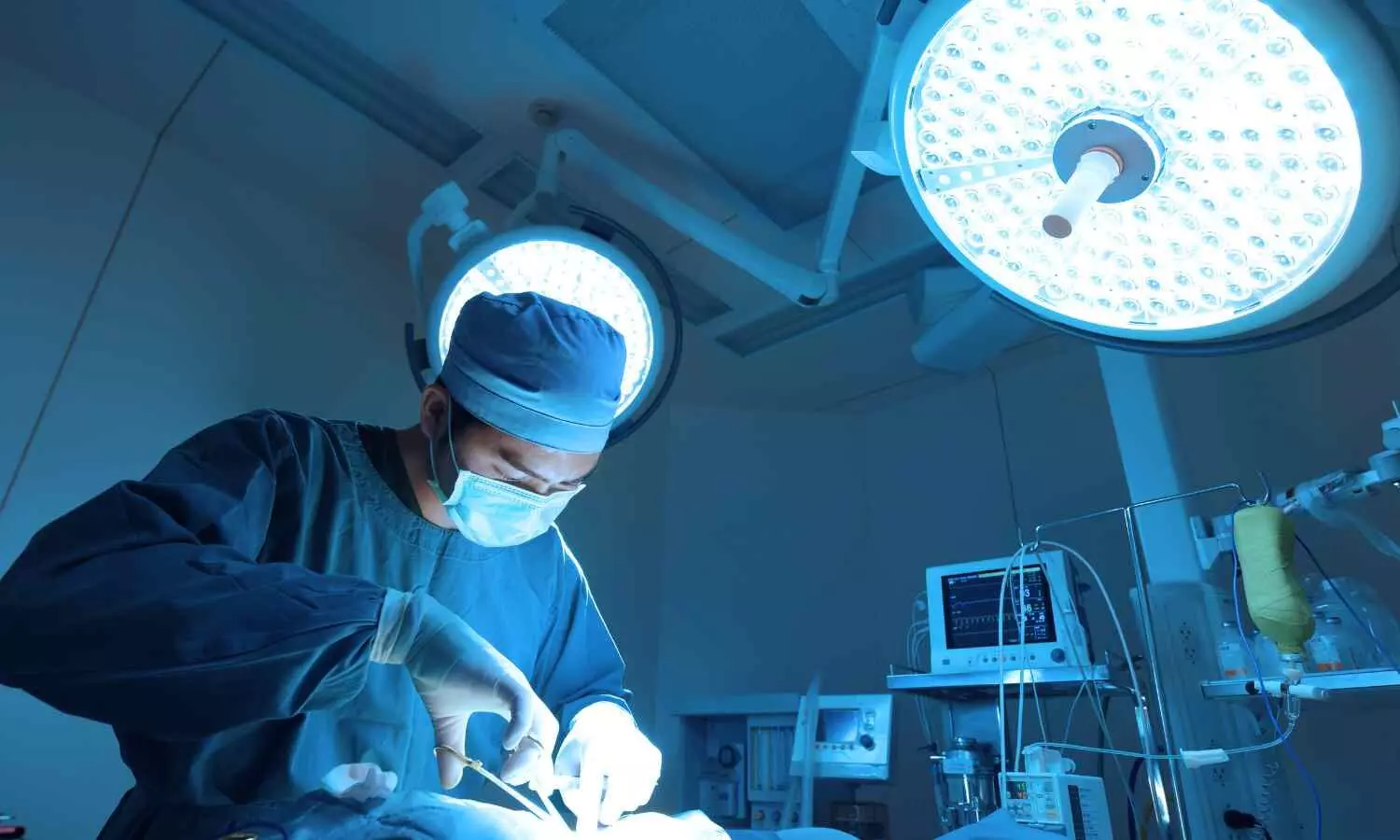
Integra LifeSciences has received 510(k) clearance from the U.S. Food & Drug Administration for its CUSA Clarity Ultrasonic Surgical Aspirator System to be used in cardiac surgeries, specifically for the debridement of unwanted tissue in procedures such as valve replacement and repair.
The system-already indicated for a range of surgical specialties like neurosurgery, orthopedic, plastic/reconstructive, thoracic, laparoscopic, gynecological, and hepatic surgeries- now expands its intended use to include cardiac surgery.
In addition to cardiac surgery, the CUSA Clarity Ultrasonic Surgical Aspirator System is indicated for use in surgical procedures where fragmentation, emulsification and aspiration of soft and hard (e.g. bone) tissue is desirable, such as neurosurgery, plastic and reconstructive surgery, orthopedic surgery, thoracic surgery, laparoscopic surgery, gynecological surgery, and liver resection and transplant surgery.
“The utilization of CUSA Clarity for cardiac surgery marks the next pivotal expansion of the ultrasonic surgical aspirator system’s breadth of indications,” said Mike McBreen, executive vice president and president of Integra’s Codman Specialty Surgical division. “As one of the leaders in the industry, this milestone strengthens our ability to deliver advanced surgical solutions to support restoring patients’ lives.”
Integra LifeSciences will spotlight the CUSA Clarity Ultrasonic Surgical Aspirator System at the American Association for Thoracic Surgeon Mitral Valve Conclave in New York City on December 11 and 12, 2025.