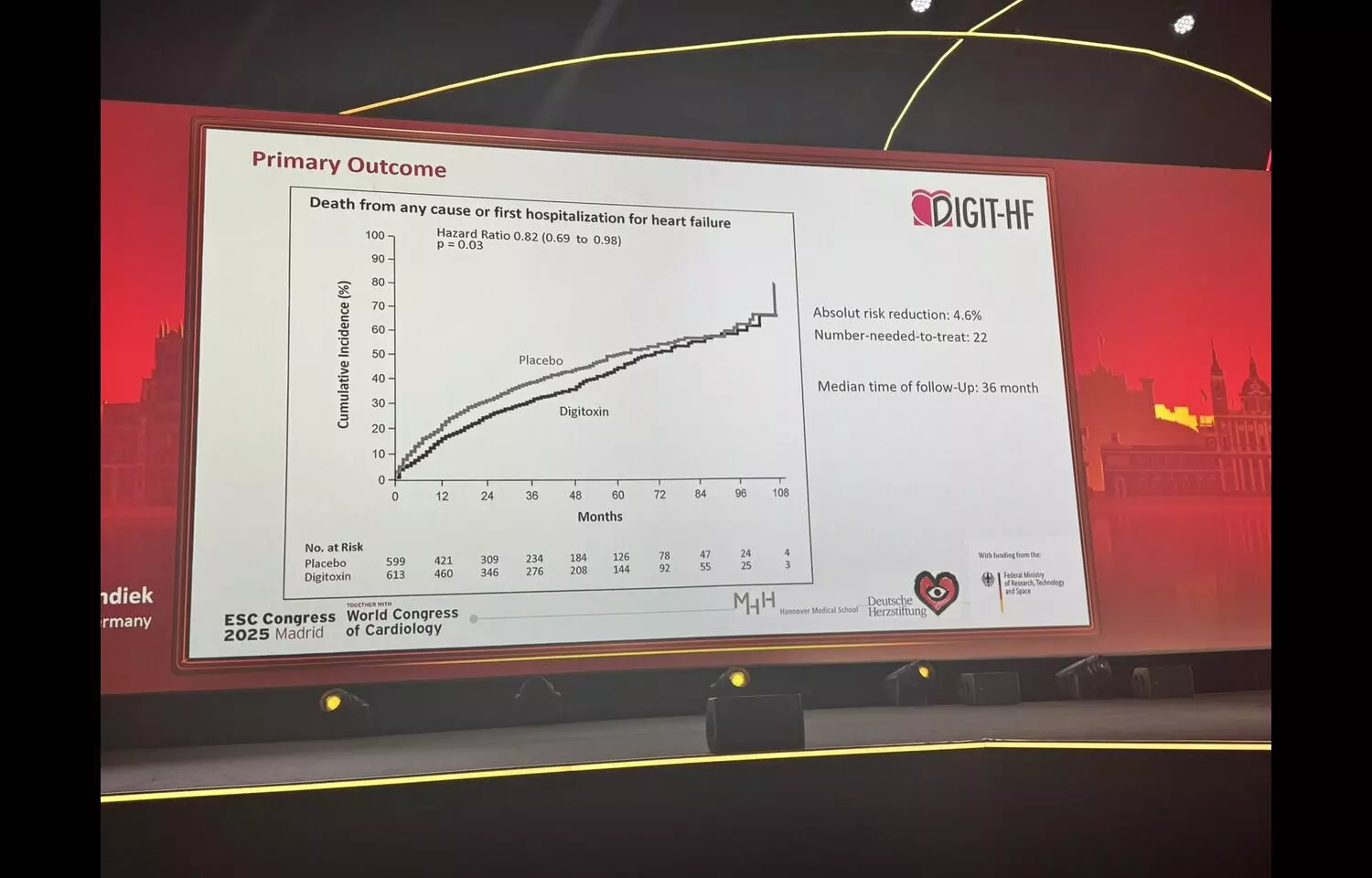
AI helped older adults report accurate blood pressure readings at home: Study
Artificial intelligence (AI) voice agents helped older adults with high blood pressure to accurately report their blood pressure readings and improved blood pressure management, according to preliminary research presented at…