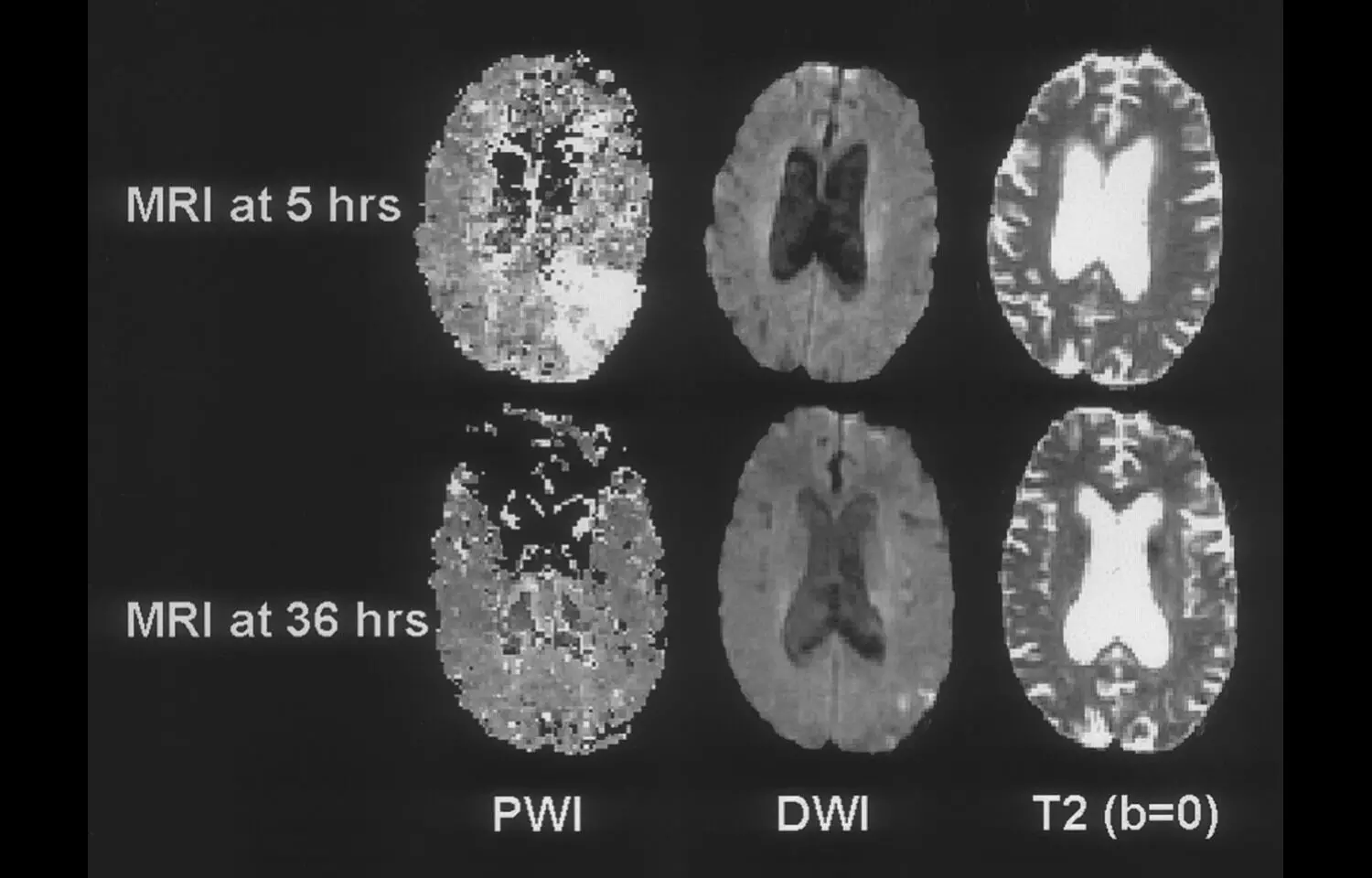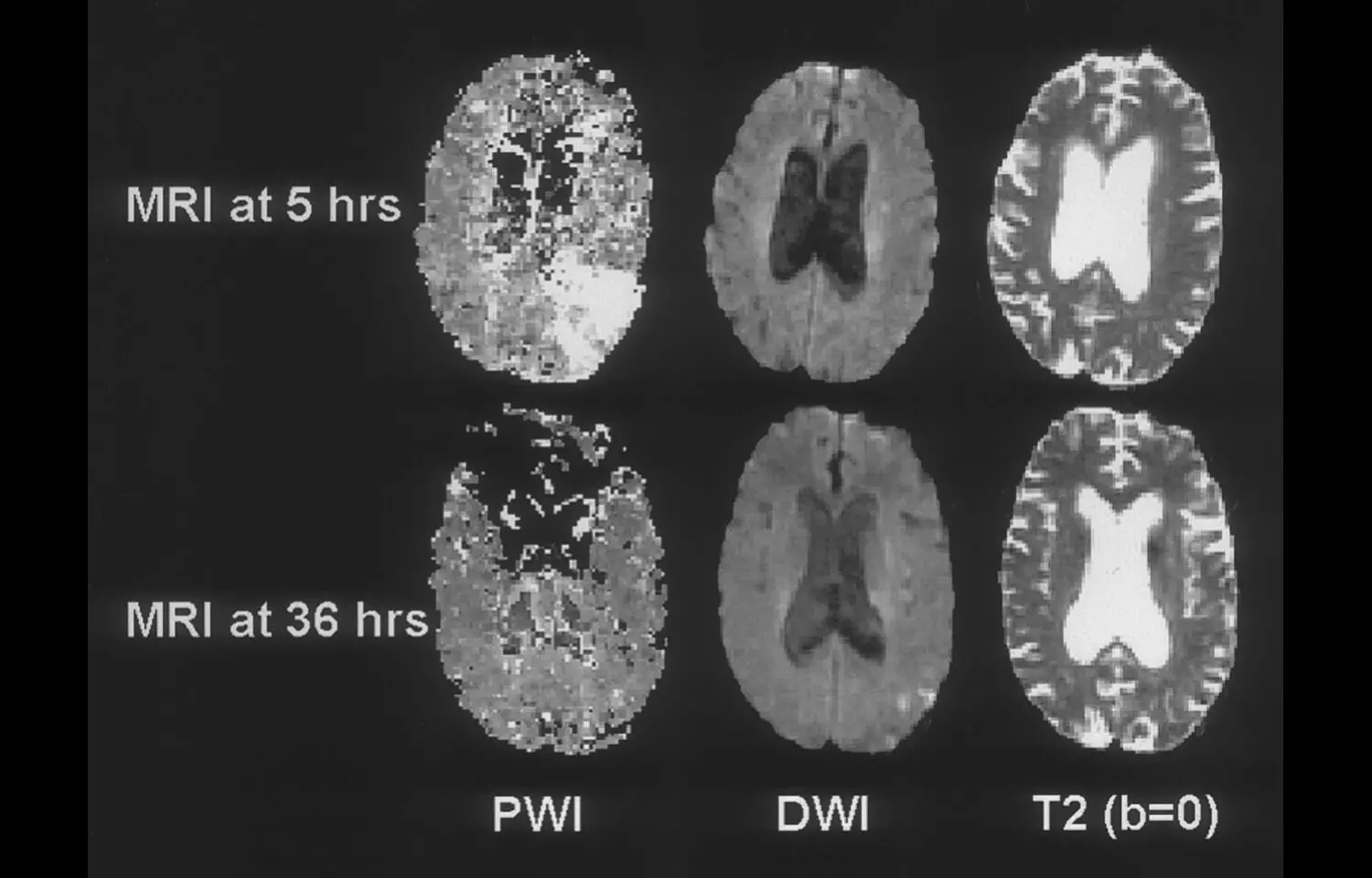
A recently published randomized, open-label clinical trial in JAMA reveals that intravenous alteplase administered 4.5 to 24 hours after acute ischemic stroke onset may significantly enhance functional recovery in patients with perfusion-imaging evidence of salvageable brain tissue, particularly when thrombectomy was not initially performed. The study enrolled 372 patients across 26 Chinese stroke centers between mid-2021 and mid-2024, all presenting beyond the standard 4.5-hour treatment window but showing imaging patterns indicating viable tissue. Results showed that 40% of patients treated with alteplase achieved functional independence (modified Rankin Scale score of 0–1) at 90 days, compared to only 26% in the control group—a statistically significant benefit (adjusted risk ratio 1.52; P = .004)PubMed. While the treatment group faced a higher rate of symptomatic intracranial hemorrhage (3.8% vs. 0.5%; adjusted risk ratio 7.34; P = .01), overall mortality at 90 days was equivalent between groups (11%)PubMed. These findings suggest that, in carefully selected patients identified via perfusion imaging, alteplase may extend its therapeutic benefit well beyond the traditional time window, offering functional gains even when endovascular options aren’t pursued. However, the elevated bleeding risk underscores the need for meticulous patient selection and monitoring. Still, this research could pave the way toward expanding thrombolytic therapy in resource-limited settings or for patients arriving late—so long as salvageable tissue is confirmed via imaging.
Keywords: alteplase, ischemic stroke, extended time window, salvageable brain tissue, perfusion imaging, JAMA, functional independence, intracranial hemorrhage, thrombolysis